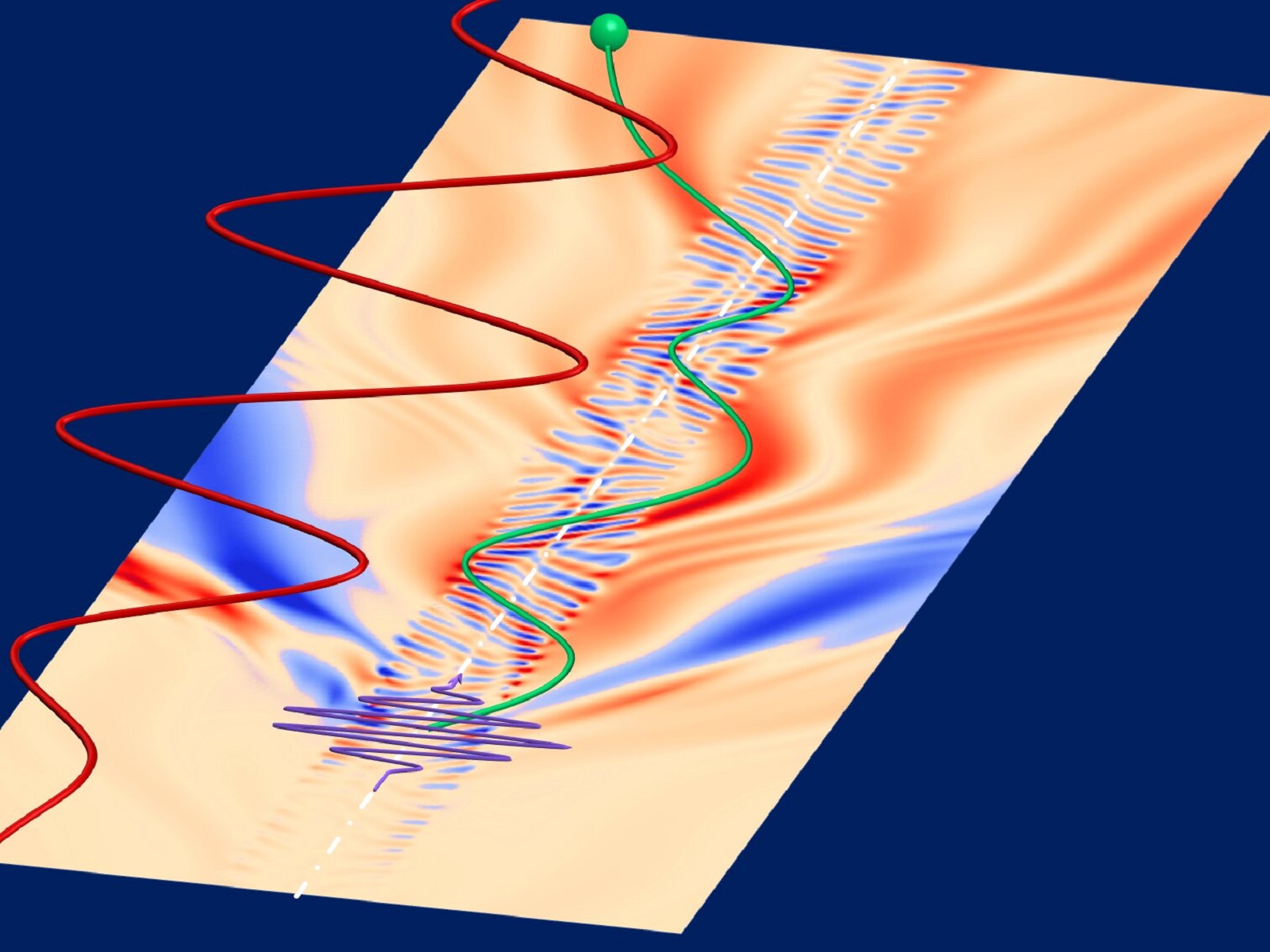
As the authors of the publication in Physical review letters, were able to combine the absorption spectrum of an ionizing pulse of intense ultraviolet light with the movement of free electrons. This motion is made possible by pulses of near-infrared light.
Read also: Quantum tricks have taken microscopy to a higher level
The conversion of optical or near infrared light into intense ultraviolet light is a very nonlinear process. In this case, the authors decided to use a short pulse of UV light, the length of which was calculated in seconds. To be precise, one attosecond is equal to one trillionth of a second. In addition, the authors were able to tune the near-infrared light in situations where tunnel ionization would be called or would be too difficult. Said ionization refers to the state from which electrons in an atom can escape after crossing a potential barrier. If an intense electric field is applied, the barrier is highly deformed.
Electron collisions have been observed with short pulses of ultraviolet light
The combination of these two aspects allowed the scientists to study re-collision of electrons. According to Ott, the approach used refers to helium as a model system and combines the absorption spectrum of ionizing light with electron trajectories. What are the benefits? First and foremost, it is about realizing the dynamics achieved with a single spectroscopic measurement without focusing separately on the time delay.
Read also: Physicists tossed a single atom and then captured it with a laser. This is an important achievement
As for the results of the observations, the researchers demonstrated, among other things, that each collision of an electron with a helium atom leads to a characteristic modification and time-dependent increase in the atomic dipole. The described method makes it possible to study the motion of electrons in an efficient way, and the authors themselves claim that it can also be applied to other systems. They mention, for example, the possibility of studying the dynamics of electrons in the company of larger atoms or molecules.

Echo Richards embodies a personality that is a delightful contradiction: a humble musicaholic who never brags about her expansive knowledge of both classic and contemporary tunes. Infuriatingly modest, one would never know from a mere conversation how deeply entrenched she is in the world of music. This passion seamlessly translates into her problem-solving skills, with Echo often drawing inspiration from melodies and rhythms. A voracious reader, she dives deep into literature, using stories to influence her own hardcore writing. Her spirited advocacy for alcohol isn’t about mere indulgence, but about celebrating life’s poignant moments.