Physicists have succeeded in measuring the difference in the magnetic properties of two highly charged isotopes of neon at a previously unavailable precision. As a result, a record-breaking quantum electrodynamics test was performed – the results were in line with the predictions of the Standard Model.
Electrons are one of the most important components of matter, and they are distinguished by specific properties, including. Spin (internal angular momentum). The spin force is determined by the so-called fission coefficient g. The fission coefficient g can be determined with great precision by quantum electrodynamics (QED), a quantum field theory that describes electromagnetic interactions. Currently Scientists from the Max Planck Institute for Nuclear Physics (MPIK) in Heidelberg They measured the g factor with an accuracy of 12 digits, which is one of the most accurate measurements in physics.
Thanks to our work, we have been able to examine QED predictions with unprecedented accuracy, and partly for the first time. To do this, we considered the difference in g-factor for two isotopes of highly charged neon ions that have only one electron. It is similar to hydrogen, but has a nuclear charge ten times greater, which enhances the effects of QED.Sven Sturm of the Max Planck Institute for Nuclear Physics, and lead research team
Notably, isotopes differ only in the number of neutrons in the nucleus, and the nuclear charge is the same. Isotopes have been studied twentyNe9+ And the 22Ne9+ With 10 and 12 neutrons respectively.
Electron trap
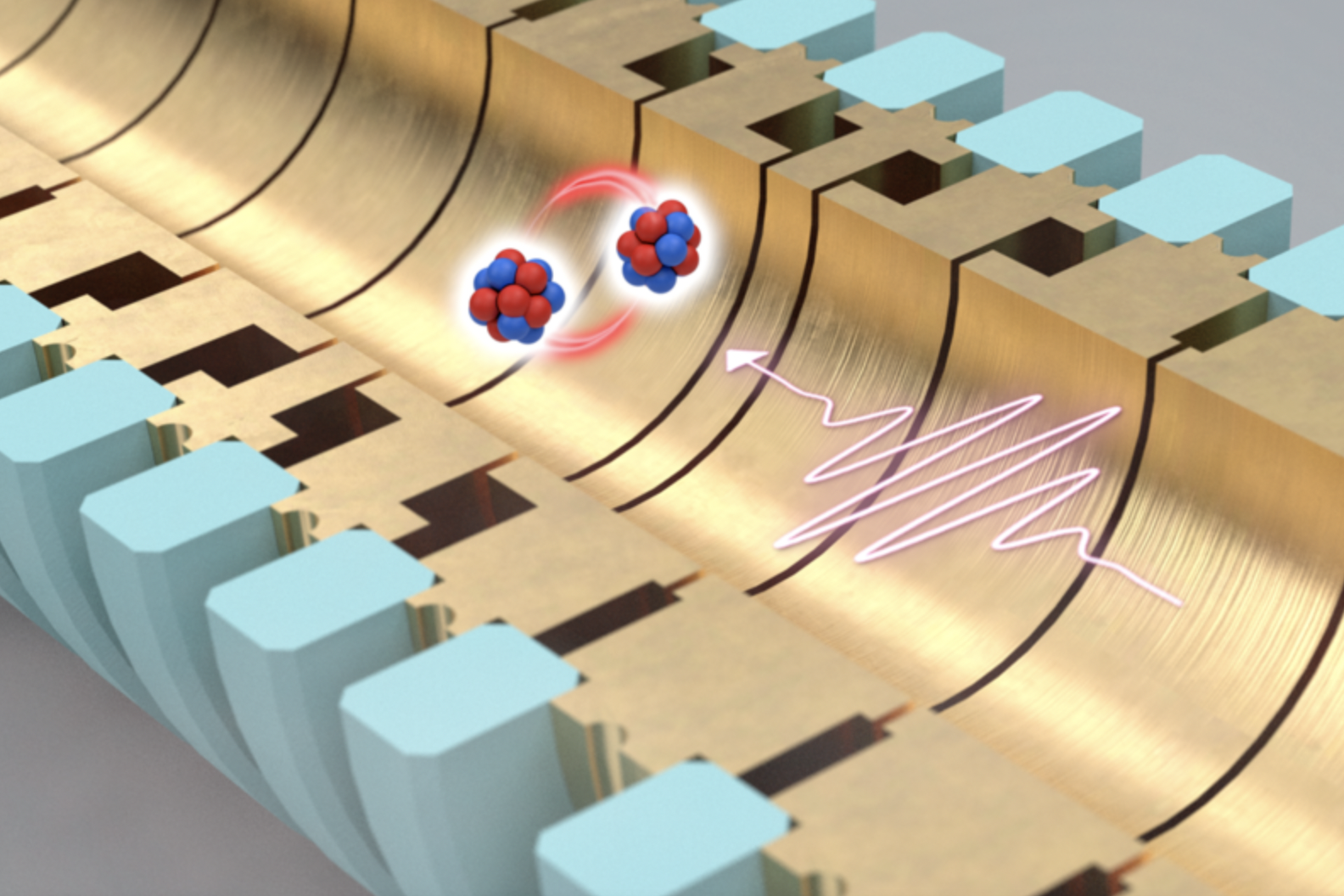
Physicists have used a specially designed Penning trap to store single ions in a strong magnetic field with 4 Tesla induction in a near-perfect vacuum. The objective of the measurement was to determine the energy required to change the direction of rotation in the magnetic field. The experiment was called ALPHATRAP.
Comparative ions – twentyNe9+ And the 22Ne9+ – They are simultaneously stored in the same magnetic field in a double motion. In such a motion, the two ions always rotate against each other in a common circular path with a radius of only 200 μm.Fabian Heiße is a PhD student in the ALPHATRAP . experience
Read also: Quantum Physics – Seven Facts You Should Know
In practice, this means that all fluctuations (small changes) in the magnetic field have an almost identical effect on both isotopes. As a result, scientists were able to determine the difference in the coefficients of both isotopes with a standard accuracy of 13 digits. Previous measurements, despite many efforts, provided an accuracy of 11 digits.
Comparing with the new experimental values, we confirmed that the electron does indeed interact with the atomic nucleus through photon exchange, as predicted by QED.Zoltan Haarmann, President of the Physicists Group
Assuming the QED results are known, the study can determine the isotope’s nuclear radius with ten times greater accuracy than previously possible.

Echo Richards embodies a personality that is a delightful contradiction: a humble musicaholic who never brags about her expansive knowledge of both classic and contemporary tunes. Infuriatingly modest, one would never know from a mere conversation how deeply entrenched she is in the world of music. This passion seamlessly translates into her problem-solving skills, with Echo often drawing inspiration from melodies and rhythms. A voracious reader, she dives deep into literature, using stories to influence her own hardcore writing. Her spirited advocacy for alcohol isn’t about mere indulgence, but about celebrating life’s poignant moments.