Scientists at Boston University School of Medicine have made an important discovery regarding nuclear pores. This could not only contribute to a better understanding of biology at its most basic level, but also contribute to new antiviral, anticancer and antiaging therapies.
Nuclear pores are openings in the double membrane of the nuclear membrane that transport molecules from the nucleus to the cytoplasm (and vice versa). Each nuclear pore consists of what is called a nuclear pore complex (NCP) with a diameter of 120-150 nm. Depending on age, metabolic activity and type, cells have a different number of pores. There are about 3000-7000 of them in hepatocytes (low metabolic activity), and in the most active cells, it reaches 50 million!
Read also: This is how cells “eat”. Scientists monitored it with high accuracy
Defects in building NPCs are associated with many diseases, including viral infections, cancer or neurodegenerative diseases.
Nuclear pores are more precise than ever before
Using cryogenic electron microscopy (cryo-EM), Researchers from Boston University School of Medicine, a yeast NPC model that revealed the complex structure of nuclear pores. This could be an important finding, also in the context of work on future treatments.
This research significantly expands our understanding of the NPC structure of brewer’s yeast, a model organism used to study the biology of nucleolar-containing cells, and thus provides new insight at many levels into the functions of this transport machine.
the professor. Christopher W. Aki Zee Boston University School of Medicine
The developed model will help to better understand how the large channels fold and change the structure to accommodate the particles being transported. The molecule is often placed in the channel like a ‘keyhole’.
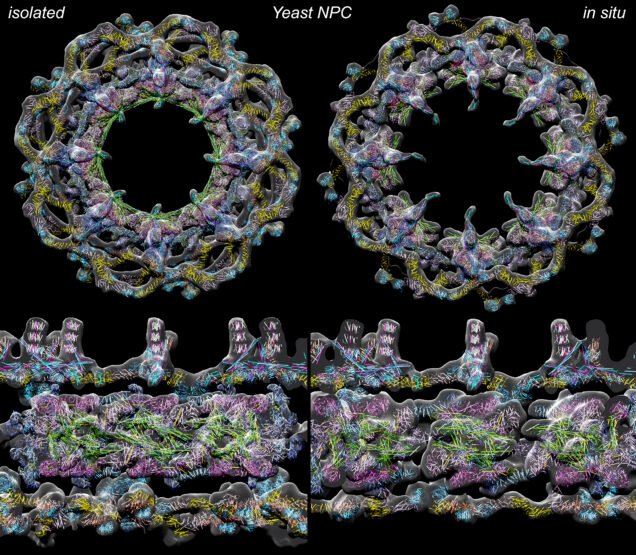
For the first time, we observed multiple types of non-playable characters in the same cell, reflecting the ability of this LEGO-like complex to use interchangeable parts to modify the structure on the nuclear side. This adaptability may play a role in adapting the functions of these machines to the different local environments in the periphery of the nucleus.
the professor. Christopher W. Aki
The results of the research were published in the journal cell. It opens the way to a better understanding of the mechanisms that viruses use to infect cells as well as changes in physiology to cause disease.

Echo Richards embodies a personality that is a delightful contradiction: a humble musicaholic who never brags about her expansive knowledge of both classic and contemporary tunes. Infuriatingly modest, one would never know from a mere conversation how deeply entrenched she is in the world of music. This passion seamlessly translates into her problem-solving skills, with Echo often drawing inspiration from melodies and rhythms. A voracious reader, she dives deep into literature, using stories to influence her own hardcore writing. Her spirited advocacy for alcohol isn’t about mere indulgence, but about celebrating life’s poignant moments.